AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
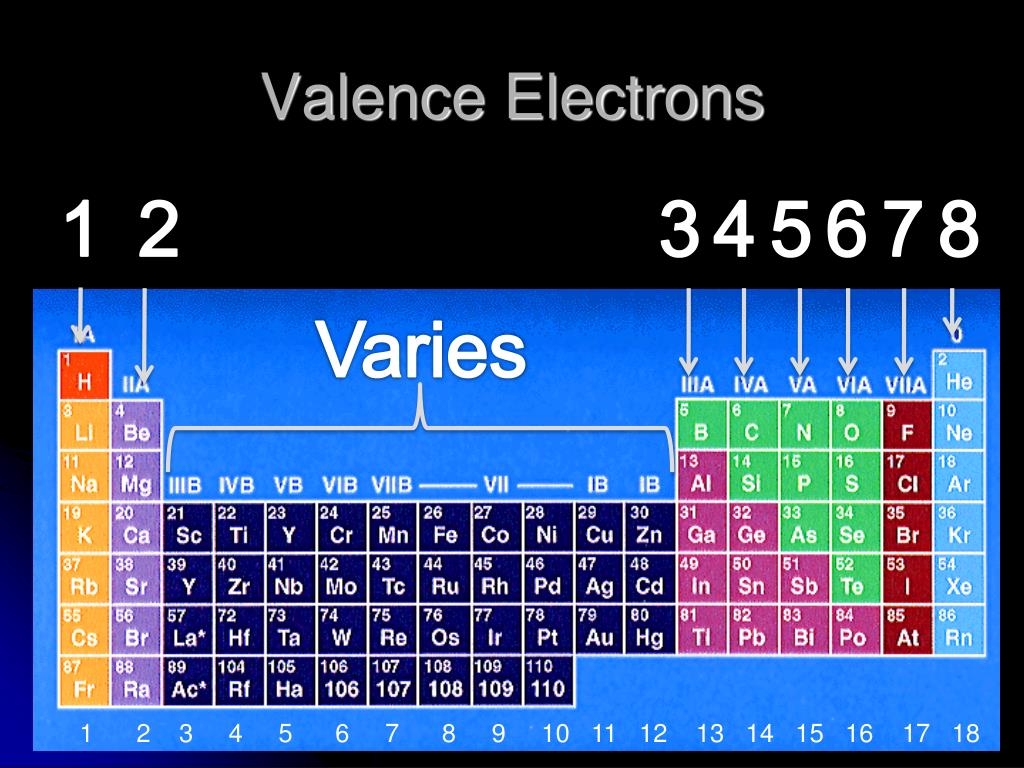
(2012, December 18) Valence Electrons and the Periodic Table. If the valence shell of an element is full, such as with a noble gas, then the element does not want to gain or lose an electron.įor example, alkali metals, which all have a valency of 1, want to lose that one electron and are likely to form ionic bonds (such as in the case of NaCl, or table salt) with a Group 17 element, which has a valency of 7 and wants to gain that one electron from the alkali metal (Group 1 element) to form a stable valency of 8.įor more on valence electrons and how they're related to the periodic table, I strongly recommend this video:Ĭitations: Tyler Dewitt. This electron arrangement indicates that the outermost orbit of Beryllium (Be) has 2 electrons. Hence beryllium element has the electrons arrangement 2, 2. There are many isotopes of chromium, out of which 52 Cr is the most abundant (around 84). The atomic mass of chromium is 51.996 u and has a density of 7.19 g/cm 3. Melting point of chromium is 1907 ☌ and its boiling point is 2671 ☌. They determine how "willing" the elements are to bond with each other to form new compounds. The atomic number of Beryllium (Be) is 4. Chromium is a strong metal having a silvery metallic lustre. Valence electrons are responsible for the reactivity of an element. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table.įor example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively.Ītoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. Valence electrons are the electrons present in the outermost shell of an atom. To form a covalent bond, one electron from the halogen and one electron from another atom form a shared pair.įor example, in #"H–F"#, the dash represents a shared pair of valence electrons, one from #"H"# and one from #"F"#. To form an ionic bond, a halogen atom can remove an electron from another atom in order to form an anion (e.g., #"F"^"-", "Cl"^"-"#, etc.). They have one less electron configuration than a noble gas, so they require only one additional valence electron gain an octet. The most reactive nonmetals are the halogens, e.g., #"F"# and #"Cl"#. The osmium element is present in rare quantities in the earth’s crust and its concentration in earth’s crust is only 1. Osmium is the most dense element present on the periodic table of elements. Nonmetals tend to attract additional valence electrons to form either ionic or covalent bonds. Osmium was discovered by Smithson Tennant in 1803 (in London). They need to lose only one or two valence electrons to form positive ions with a noble gas configuration. The most reactive metals are those from Groups 1 and 2. While these are the most common valences, the real behavior of electrons is less simple. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in #ns^2 np^6#. You may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups (columns) of the periodic table. Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
